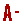
As the world population increases, water has become a crucial resource for domestic, commercial and industrial uses. These activities can lead to increased levels of contaminants being discharged. Various types of contaminants are found in water bodies, including endocrine-disrupting compounds (EDCs) (Schuwirth et al., 2018). Most EDCs originate from consumer products such as pesticides, personal care products, plastics and detergents (Liu et al., 2019a, Liu et al., 2019b, Liu et al., 2019c). These compounds are chemically active and can harm humans and animals by impacting the central nervous system and reproductive system (Wee and Aris, 2017). Even at trace levels, EDCs have a strong effect on the endocrine system and can bioaccumulate in the body (Ismail et al., 2017; Jun et al., 2019; Puma et al., 2010). EDCs constitute a wide range of both natural (e.g., hormones) and synthetic (e.g., industrial by-products) exogenous and emerging chemicals (Wee and Aris, 2017). EDCs are released into the environment through sewage systems and can then be taken up by organisms (Ismail et al., 2017). Fig. 1 depicts the sources and transport of EDCs through the environment before being taken up by humans. Numerous types of EDCs (Table 1) have been detected in wastewater treatment plant discharge and water bodies, such as pharmaceuticals and medical drugs (e.g. propranolol, ibuprofen and diclofenac), steroid hormones (e.g. testosterone, progesterone, 17β-estradiol and 17α-ethynylestradiol), polyhalogenated compounds (e.g. perfluorooctanoic acid and perfluorooctanesulfonic acid) and phenolic compounds (e.g. bisphenol A, bisphenol S and bisphenol F). These compounds are difficult to remove due to their hydrophobic characteristic and resistance to biodegradation (Jun et al., 2019; Joseph et al., 2019; Aris et al., 2014; Zuo et al., 2013).
Water treatment plants play a vital role in controlling the fate and transport of EDCs in the aquatic environment. These compounds can easily flow through the treatment system and are discharged into the environment without being removed by the treatment process (Can et al., 2014). Therefore, it is urgent to monitor the concentration of EDCs in the water system. Analytical instrumentation is often applied to detect EDCs present in water samples upon discharge (Kasonga et al., 2021; Basile et al., 2011), and treatment plants can remove a substantial amount of EDCs. However, treatment processes are unable to eliminate low concentrations of EDCs in water (Gadupudi et al., 2019). To remove EDCs from the water system, several types of treatment methods have been considered, for example, advanced oxidation, ozonation, electrochemical oxidation and photocatalytic decomposition (Jun et al., 2019). Bertanza et al. (2010) applied an advanced oxidation process of UV/H2O2 and ozonation to treat effluent and eliminated several phenolic compounds; however, the process showed only approximately 70% removal. These methods are not amenable for treating industrial wastewater, as the high concentrations of contaminants would lead to high costs and consume a large amount of energy.
These advanced methods are too expensive because of high operational and capital costs (Guerra et al., 2015). As an alternative, adsorption-based treatment has been reported as the most suitable method for EDC removal, as this method is inexpensive and more efficient (Fig. 2) (Lim and Aris, 2014). Moreover, adsorption materials have been widely used in water and wastewater treatment due to their strong interactions with hydrophobic organic contaminants such as EDCs (Joseph et al., 2019). Adsorbents used in adsorption treatment can be either natural organic, natural inorganic, or synthetic materials (Ndimele et al., 2018). Metal-organic frameworks (MOFs) are synthetic materials that have received considerable attention due to their useful characteristics such as diverse geometries, high porosity, thermal stability, selectivity and recyclability (Sun et al., 2019; Kadhom and Deng, 2018; Li et al., 2018a, ; Aris et al., 2020; Jun et al., 2019). MOFs are metal–ligand coordination polymers constructed by a covalent bond between metals clusters and organic ligands, forming various porous frameworks such as one-dimensional (1D) chains, two-dimensional (2D) interpenetrating layers and three-dimensional (3D) materials (Alhamami et al., 2014; He et al., 2010). A range of structural, magnetic, electrical, optical and catalytic properties can be incorporated through the choice of metal cluster and organic linker (Razavi and Morsali, 2019). Unlike other porous materials, a typical crystalline MOF structure has a porosity of over 50%, and the surface areas of MOFs range between 1000 and 10,000 m2/g. These values are larger than those of typical porous materials such as carbons and zeolites (Furukawa et al., 2013). Thus, MOFs can be used in specific applications, especially for gas capture, drug delivery, as catalysts and sensors, in separation/purification and as electrical conductors (Sun et al., 2020; Dhakshinamoorthy et al., 2011; Lazaro and Forgan, 2019; Mahata et al., 2017; Pi et al., 2018; Sun et al., 2016). Many studies have exploited the ability of MOFs to adsorb contaminants in aqueous media. A material that is workable in an aqueous medium should ideally meet the following seven criteria: (i) high adsorption capacity; (ii) high selectivity; (iii) fast adsorption kinetics; (iv) regeneration ability; (v) long-term stability; (vi) tolerance to the other compounds; and (vii) low production cost (Abdelnaby et al., 2018).
READ MORE --→
Fahren Fazzer Sukatis, Sze Yee Wee & Ahmad Zaharin Aris
Department of Environment, Faculty of Forestry and Environment, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia
* You can continue reading the article by clicking the button (READ MORE) above